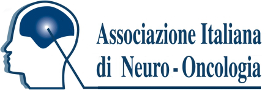Rebound Growth Following Temporary Discontinuation of BRAFi and MEKi in a Patient With Multifocal BRAFV600E-Mutant Primary Brain Tumor
Antonio Pisano, 1 Samanta Cupini, 2 Orazio Santo Santonocito, 1 Bianca Pollo, 3 Rosina Paterra, 4 Giacomo Allegrini, 2 Chiara Finale, 2 Francesca Vannozzi, 5 Claudia Scudieri, 1 Vita Maria Montemurro, 1 Elena Anghileri, 4 and Anna Luisa Di Stefano 1, 6
Neurology® 2025;104:e213336. doi:10.1212/WNL.0000000000213336
Correspondence: Dr. Di Stefano - annaluisa.distefano@uslnordovest.toscana.it
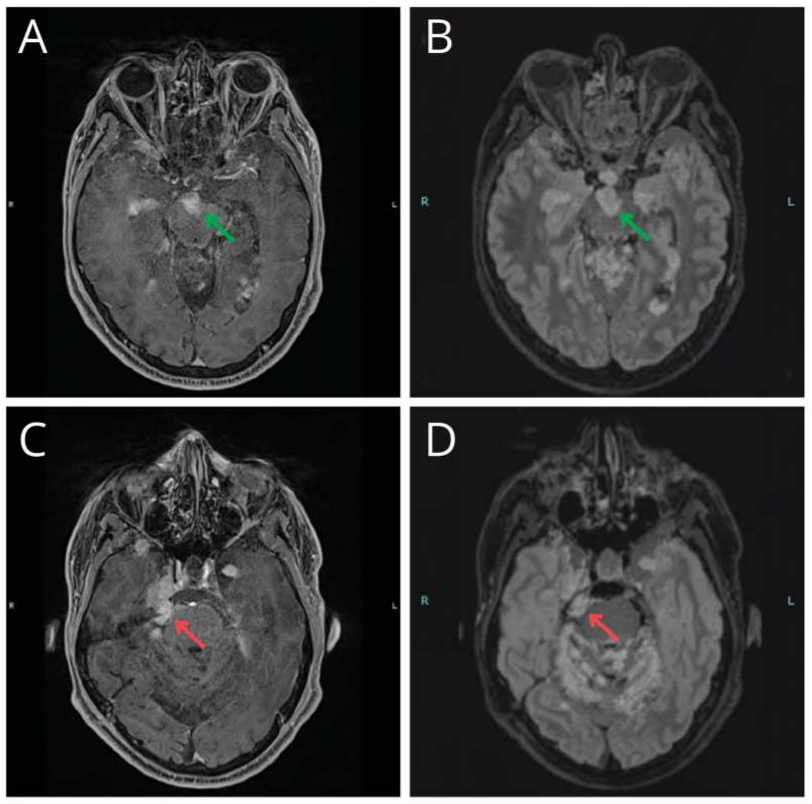
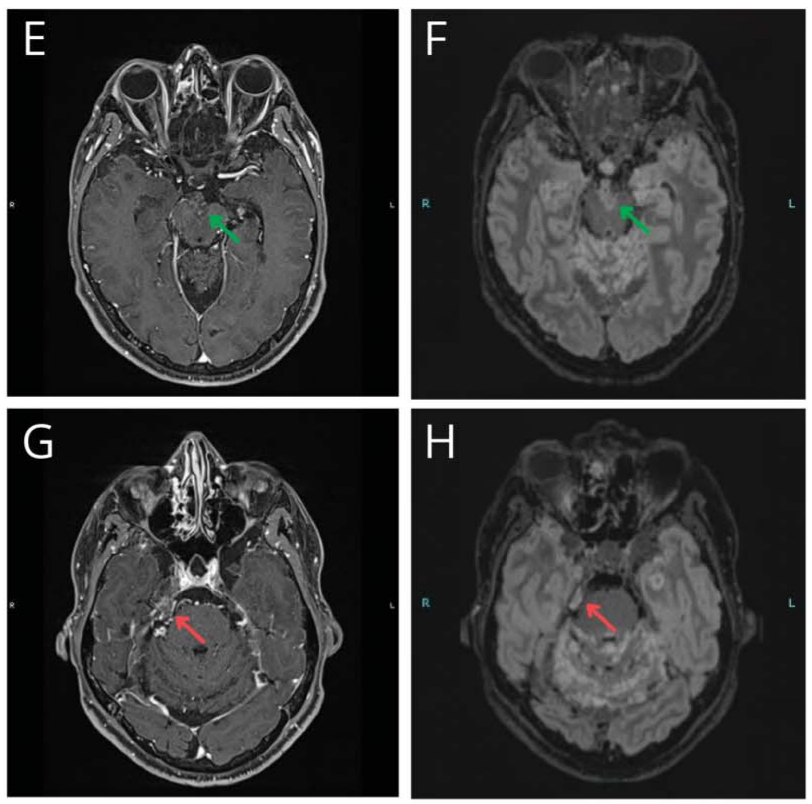
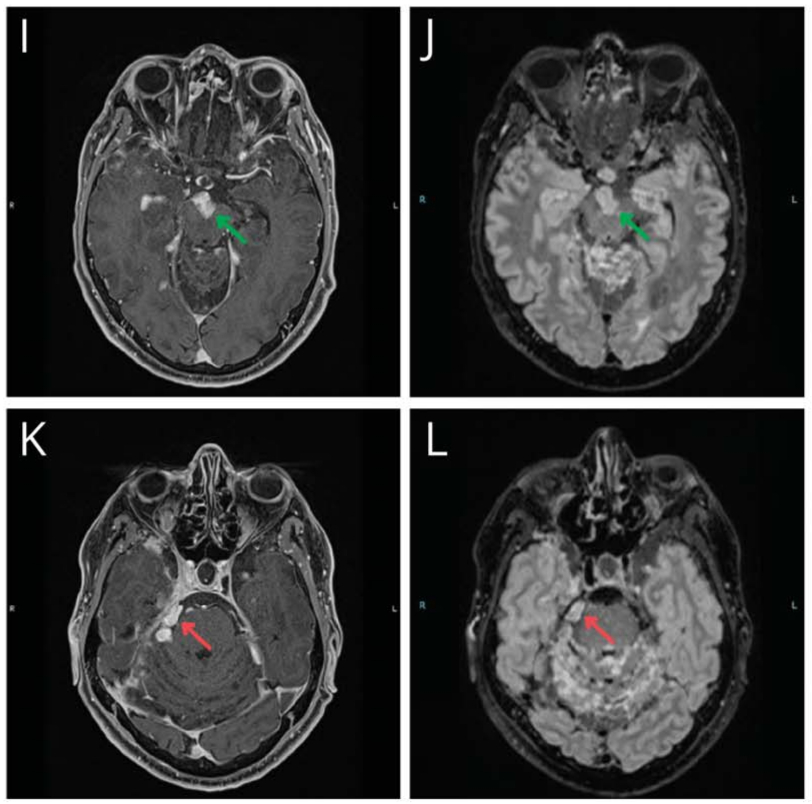
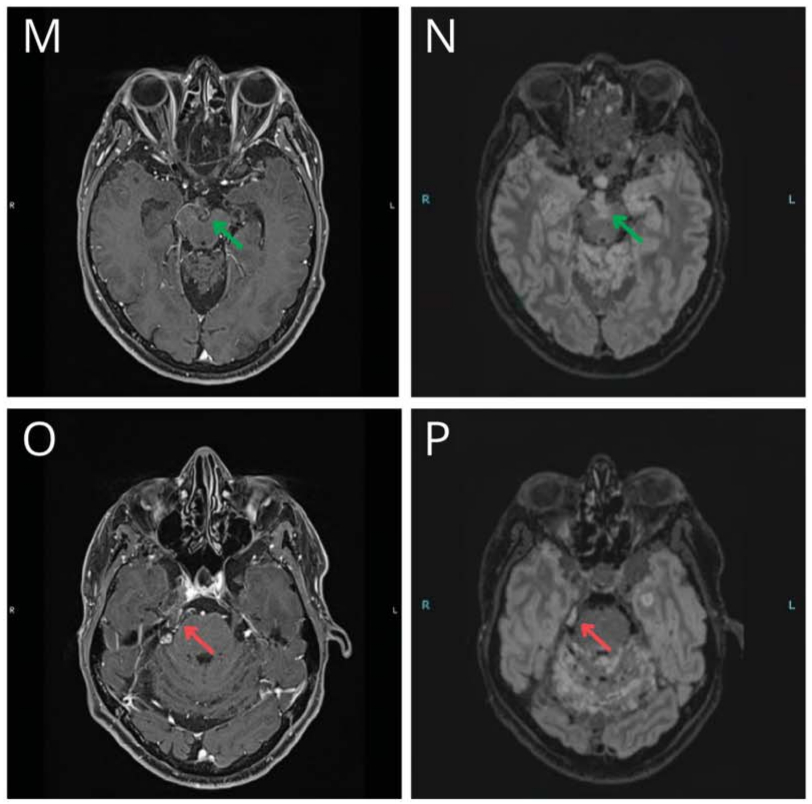
We present the case of a 47-year-old man with a progressing BRAFV600E-mutant diffuse leptomeningeal glioneuronal tumor (DLGNT) treated with dabrafenib and trametinib.1 At age 24, he underwent surgical excision of a third ventricle lesion, followed by surveillance, craniospinal irradiation, and chemotherapy at age 35, as well as radiosurgery for subsequent recurrences. At age 45, BRAFV600E mutant detection led to targeted therapy with dabrafenib and trametinib (Video 1A). After 4 cycles, imaging showed a “minor” response (Video 1B; Figure, E–H) with clinical improvement.2 After a 1-week therapy interruption during cycle 6 due to a mild skin rash, tumor lesions increased in size (Video 1C; Figure, I–L) without clinical changes. Subsequent MRI showed improvement after resuming therapy (Video 1D; Figure, M–P), which persists through 17 cycles. The patient did not receive steroids or bevacizumab during targeted therapy. This case illustrates possible “rebound growth” from temporary therapy interruption.
Author Contributions
A. Pisano: drafting/revision of the manuscript for content, including medical writing for content; major role in the acquisition of data; analysis or interpretation of data. S. Cupini: drafting/revision of the manuscript for content, including medical writing for content; major role in the acquisition of data; analysis or interpretation of data. O.S. Santonocito: major role in the acquisition of data; analysis or interpretation of data. B. Pollo: major role in the acquisition of data; analysis or interpretation of data. R. Paterra: major role in the acquisition of data; analysis or interpretation of data. G. Allegrini: major role in the acquisition of data; analysis or interpretation of data. C. Finale: major role in the acquisition of data; analysis or interpretation of data. F. Vannozzi: major role in the acquisition of data; analysis or interpretation of data. C. Scudieri: major role in the acquisition of data; analysis or interpretation of data. V.M. Montemurro: major role in the acquisition of data; analysis or interpretation of data. E. Anghileri: drafting/revision of the manuscript for content, including medical writing for content; major role in the acquisition of data; analysis or interpretation of data. A.L. Di Stefano: drafting/revision of the manuscript for content, including medical writing for content; major role in the acquisition of data; study concept or design; analysis or interpretation of data.Acknowledgment
ALDS received support from a donation in the memory of Mr. Leonardo Viviani to Fondazione Faro Onlus.Study Funding
The authors report no targeted funding.Disclosure
The authors report no relevant disclosures. Go to Neurology.org/N for full disclosures.Figure Representative Variations in Selected Tumor Locations on BRAFi and MEKi Therapy in a Patient With a Progressing BRAFV600E-Mutant DLGNT
Publication History
Received by Neurology September 6, 2024. Accepted in final form December 2, 2024. Submitted and externally peer reviewed. The handling editor was Editor-in-Chief Jos´e Merino, MD, MPhil, FAAN.References
- Central Nervous System TumoursWHOClassification of Tumours, 5th Edition, Volume 6, IARC 2021
- Wen PY, van den Bent M, Youssef G, et al. RANO 2.0: update to the response assessment in neuro-oncology criteria for high- and low-grade gliomas in adults. J Clin Oncol. 2023;41(33):5187-5199. doi:10.1200/JCO.23.01059